IDEA Launches Push-of-a-Button Cell Imaging System
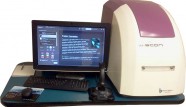
IDEA Bio-Medical Ltd. (IDEA Bio) and The Medical University of South Carolina (MUSC) announced last month that they have completed the sale of IDEA Bio’s Hermes WiScan® system to MUSC’s Center for Drug Discovery.
Hermes WiScan® is a breakthrough biological cell imaging desktop system that enables researchers, at all levels, to run a wide variety of biological applications at the push-of-a-button, using an extremely easy-to-use (tablet-style) touchscreen mechanism. The company offers a base system that meets most research needs, and an additional set of modular packages like Live Cell, Object Mapping, and more. WiScan® technology originated at Israel’s Weizmann Institute of Science, and was further developed by IDEA Bio-Medical’s parent company as part of a commercialization agreement with YEDA – the Institute’s technology transfer arm. The parent company, IDEA Machine Development, is an established Original Design Manufacturer that specializes in precision optics and laser-based technologies, and boasts such clients as KLA Tencor, HP, Orbotech, and the Israeli defense establishment.
IDEA Bio’s connection to the Southeast began with a meeting between its CEO, Shlomo Turgeman, and Tom Glaser, President of the Atlanta-based American-Israel Chamber of Commerce (AICC) during one of his visits to Israel. Shlomo later on hired Friedman, who arranged the meeting, to handle global sales and interact with distributors worldwide. “My first order of business was to sign up IDEA Bio-Medical as a member of the Chamber, and to ask for its assistance in reaching Southeastern-based universities that conduct biomedical research. Tom Glaser introduced me to several of his contacts at universities in Georgia, Alabama and South Carolina, and I soon had meetings lined up to discuss our system,” said Friedman.
“Having been involved with the Chamber, I was already aware of the amazing technologies coming out of Israel. This is why I was curious to hear more about this system,” said Stephen Lanier, Ph.D., Associate Provost for Research MUSC. He then turned it over to Patrick M. Woster, who is the South Carolina SmartState™ Endowed Chair for Drug Discovery at MUSC and Director of the Drug Design and Synthesis core facility, to test the system. “Hermes is about the size of a large printer, processes information with incredible speed, and performs a wide variety of cell-based experiments that meet our overall research needs,” said Woster.
Ultimately, the decision to purchase Hermes was of course based on the technology and the need. But the path, which led to MUSC’s awareness of the system, involved building upon relationships between people on both sides of the ocean. Back in February 2011, the AICC organized the South Carolina-Israel Collaboration initiative with a grant support from Charleston-based The InterTech Group. The collaboration is designed to foster mutual relationships between companies in Israel and in South Carolina, and their counterpart investors, partners and customers. It is focused on six clusters, among them Biomedical, and includes educational events and business exchanges around companies and academia, and the organization of business and academic delegations. In mid-November, the Chamber, together with Jonathan Zucker, President of Charleston-based InterTech Group, and Chairman of the collaboration, led a 26-person delegation to Israel. The group, which was split between business people and academic researchers, among them Lanier, got a chance to see the system in action at the Weizmann Institute. “We’re committed to doing business with Israeli-based companies, not only because of our strong connection to the country, but also because we recognize that so many advanced solutions are created in Israel. IDEA’s product is an example of how Israeli ingenuity could produce a product that works better, faster, and more reliably than its competition, yet costs a fraction of it,” said Zucker.
“Of course we have competition,” says Turgeman, “but through the help of our American friends, we were able to demonstrate how a small company from Israel could compete successfully in the large American market. We expect many more sales to follow, not only to universities but also to pharmaceutical companies, and are placing a lot of effort in building on this momentum,” he added.
Hermes WiScan® is currently undergoing onsite evaluations by pharmaceutical companies and research institutions in the Northeast and in the West Coast of the United States. In addition, the company is actively pursuing distribution agreements across the globe. Just yesterday, IDEA Bio signed an agreement with BI Biological Industries, based in Kibbutz Beit-Haemek in northern Israel, to distribute Hermes WiScan® to research institutions, pharmaceutical companies and hospitals in Israel. “”Biological Industries places great importance on scientific and business collaboration with new Israeli life science companies whose activities intersect with BI main sphere, namely tissue culture,” said Alon Ariel, CEO of Biological Industries. The company is also in advanced discussions with potential distributors in France, Spain, Holland, Canada and elsewhere around the globe.
IDEA Bio-Medical was established in 2007 as a spin-off from IDEA Machine Development – a 17-year-old Original Design Manufacturer that specializes in the manufacture of precision optics and laser-based technologies in the fields of defense, semiconductors, PCB, Flat Panel Displays, digital printing, and gemology. The company boasts such clients as HP, KLA-Tencor, Elbit Systems, Orbotech, Lazar Kaplan and others. IDEA Bio-Medical is an outcome of a commercialization agreement between its parent company and Yeda – the technology transfer arm of the Weizmann Institute of Science, which led to the development of revolutionary High-Definition, Automated Cell Imaging systems called WiScan® and an Image Analysis Platform called WiSoft®. WiScan®, comprised of two products: Hermes and Argus, combines two screening strategies: High Content, and High Throughput. Together with WiSoft®, the system provides an advanced rapid harvesting tool of complex biomedical information from primary and secondary screens and shows major improvements of up to 20 times in screening rates and screening set-up.

